NOTE: The previous post in this series is found here.
While prior posts in this series have focused on age-specific death rates and specifically on how the 2021 death rates are too high to be due to COVID, this post focuses on the shenanigans employed by the World Health Organization in a study meant to undercut the benefit of hydroxychloroquine (HCQ).
While watching an episode of The Illusion of Consensus with Dr. Pierre Kory being interviewed, it was brought out by Dr. Kory that the Solidarity trial of HCQ is borderline-criminal, due to what appears to be intentional over-dosing. In that trial HCQ was “discontinued for futility” on 19 June 2020.
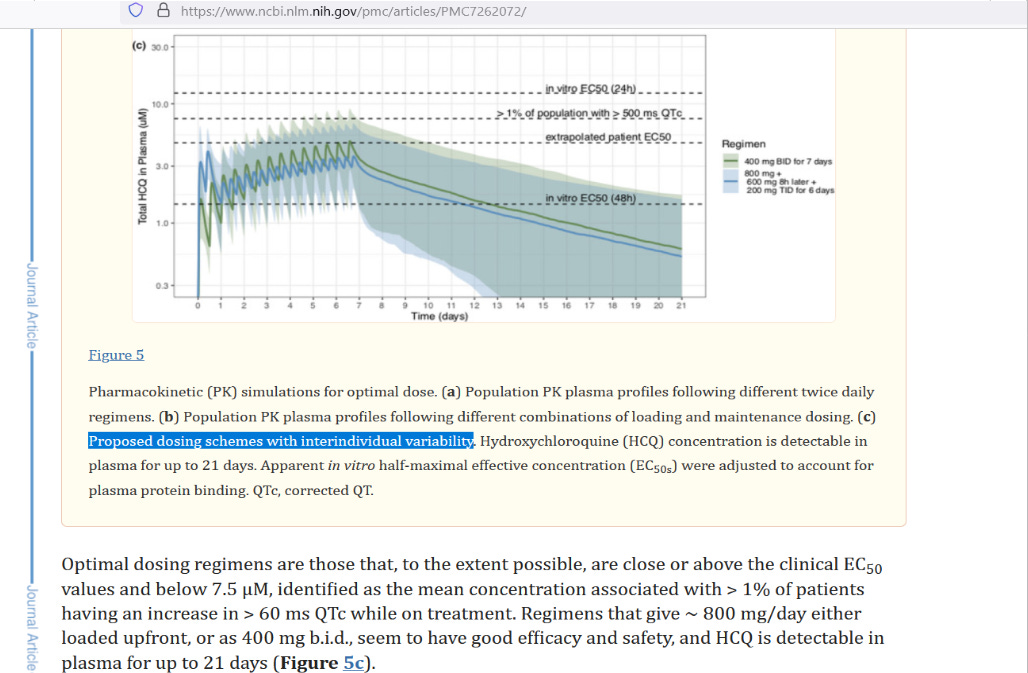
But on 12 May 2020, a key study was published electronically, introducing a suggested protocol for HCQ dosing meant to get good results without causing heart issues (a potential side effect of HCQ). Here are the two suggested protocols from that study which incorporated individual variability in order to keep people safe:
[click to enlarge]
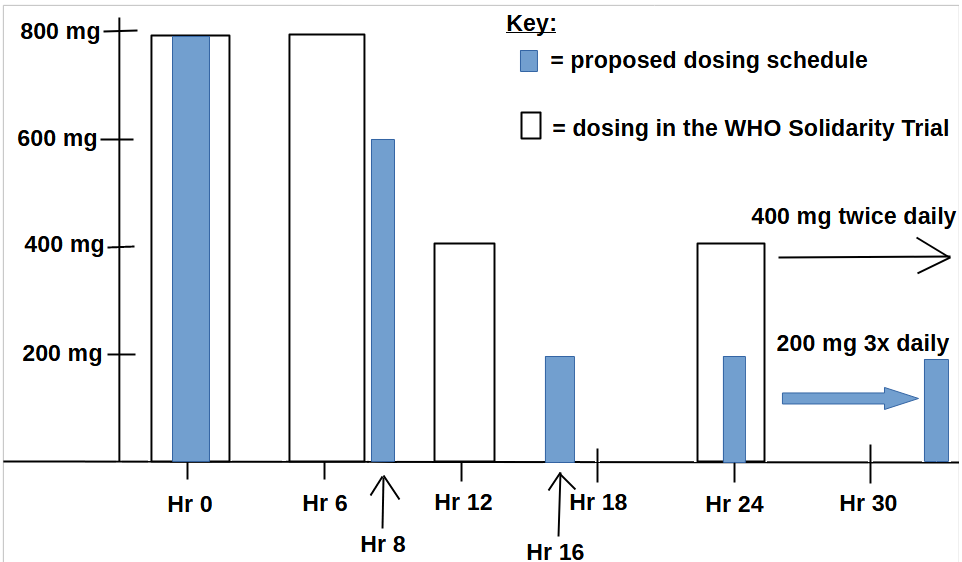
At right you have two protocols listed. The green line with lighter green shading around it is 400 mg HCQ, twice daily (800 mg daily). The blue line with lighter blue shading around it is the proposed loading dose scheme. To contrast this second dosing proposal to what WHO used in the Solidarity trial, here is an overlay:
While both regimens gave 800 mg of HCQ at Hour 0, they diverge by Hour 6. By Hour 24, the cumulative dose in the proposed dosing schedule reached 1,800 mg of HCQ, but had become titrated down to 600 mg of daily dosing (200 x 3). But those in the Solidarity trial had 2,400 mg by Hour 24, and 800 mg of daily dosing (400 x 2).
Evidence suggests that the WHO overdosed people with HCQ, which not only leads to heart problems, but which can also cause methemoglobinemia which harms oxygen delivery to tissues, thus exacerbating the low oxygen that COVID patients might have had — which would explain the higher mortality found in the HCQ recipients.
Reference
[HCQ has been associated with methemoglobinemia] — Naymagon L, Berwick S, Kessler A, Lancman G, Gidwani U, Troy K. The emergence of methemoglobinemia amidst the COVID-19 pandemic. Am J Hematol. 2020 Aug;95(8):E196-E197. doi: 10.1002/ajh.25868. Epub 2020 Jun 3. PMID: 32413176; PMCID: PMC7276830. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7276830/
[proposed loading dose for HCQ] — Garcia-Cremades M, Solans BP, Hughes E, Ernest JP, Wallender E, Aweeka F, Luetkemeyer AF, Savic RM. Optimizing Hydroxychloroquine Dosing for Patients With COVID-19: An Integrative Modeling Approach for Effective Drug Repurposing. Clin Pharmacol Ther. 2020 Aug;108(2):253-263. doi: 10.1002/cpt.1856. Epub 2020 May 12. PMID: 32285930; PMCID: PMC7262072. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7262072/
[actual loading dose used by the WHO in their Solidarity trial] — WHO Solidarity Trial Consortium; Pan H, Peto R, Henao-Restrepo AM, Preziosi MP, Sathiyamoorthy V, Abdool Karim Q, Alejandria MM, Hernández García C, Kieny MP, Malekzadeh R, Murthy S, Reddy KS, Roses Periago M, Abi Hanna P, Ader F, Al-Bader AM, Alhasawi A, Allum E, Alotaibi A, Alvarez-Moreno CA, Appadoo S, Asiri A, Aukrust P, Barratt-Due A, Bellani S, Branca M, Cappel-Porter HBC, Cerrato N, Chow TS, Como N, Eustace J, García PJ, Godbole S, Gotuzzo E, Griskevicius L, Hamra R, Hassan M, Hassany M, Hutton D, Irmansyah I, Jancoriene L, Kirwan J, Kumar S, Lennon P, Lopardo G, Lydon P, Magrini N, Maguire T, Manevska S, Manuel O, McGinty S, Medina MT, Mesa Rubio ML, Miranda-Montoya MC, Nel J, Nunes EP, Perola M, Portolés A, Rasmin MR, Raza A, Rees H, Reges PPS, Rogers CA, Salami K, Salvadori MI, Sinani N, Sterne JAC, Stevanovikj M, Tacconelli E, Tikkinen KAO, Trelle S, Zaid H, Røttingen JA, Swaminathan S. Repurposed Antiviral Drugs for Covid-19 - Interim WHO Solidarity Trial Results. N Engl J Med. 2021 Feb 11;384(6):497-511. doi: 10.1056/NEJMoa2023184. Epub 2020 Dec 2. PMID: 33264556; PMCID: PMC7727327. https://pubmed.ncbi.nlm.nih.gov/33264556/