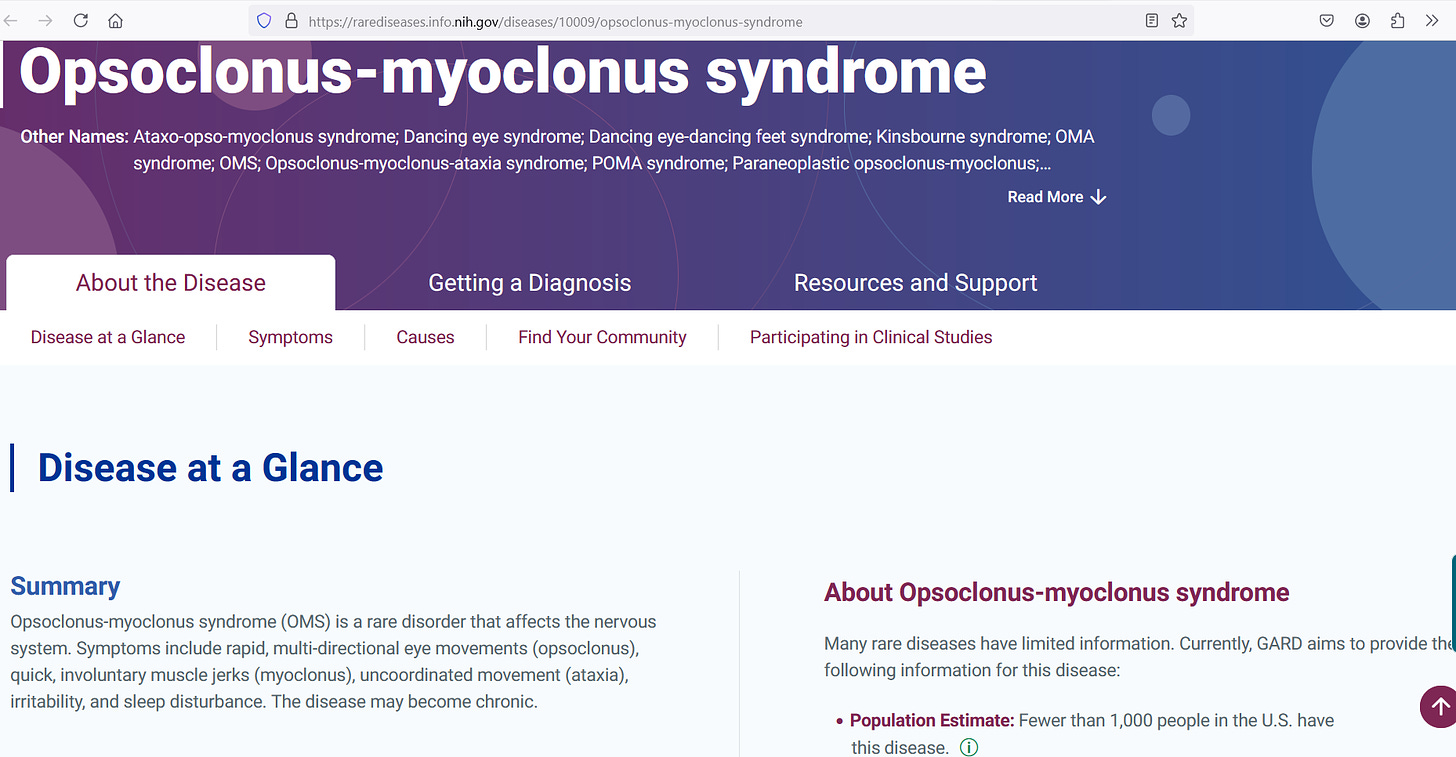
Opsoclonus myoclonus is a rare disease of the nervous system that can cause the eyes to move erratically (opsoclonus) and the muscles to twitch erratically (myoclonus) or to relax erratically (ataxia). Being such a rare disorder, not even a thousand people have it in the entire United States:
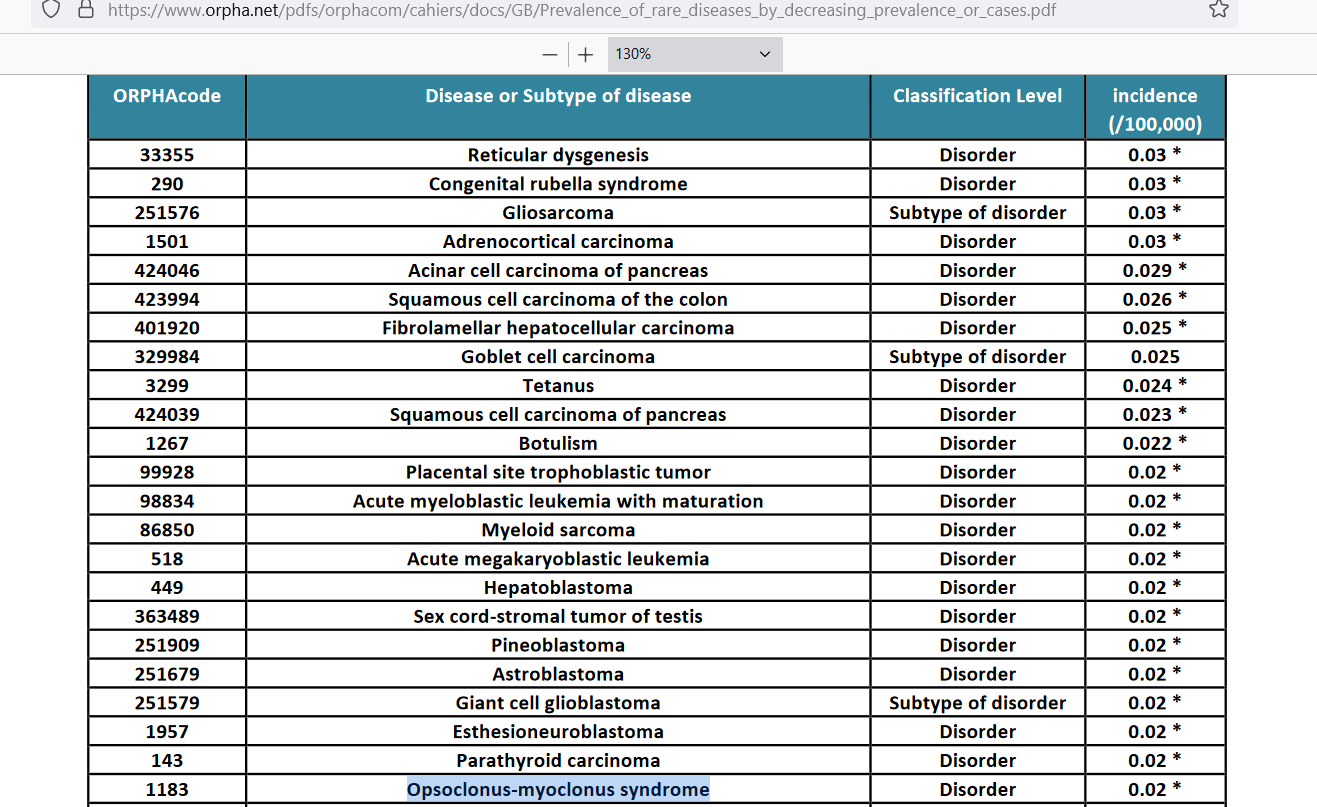
The annual incidence rate has been estimated to lie between 1 case per 10 million (0.01 per 100,000) and 1 case per 5 million (0.02 per 100,000):
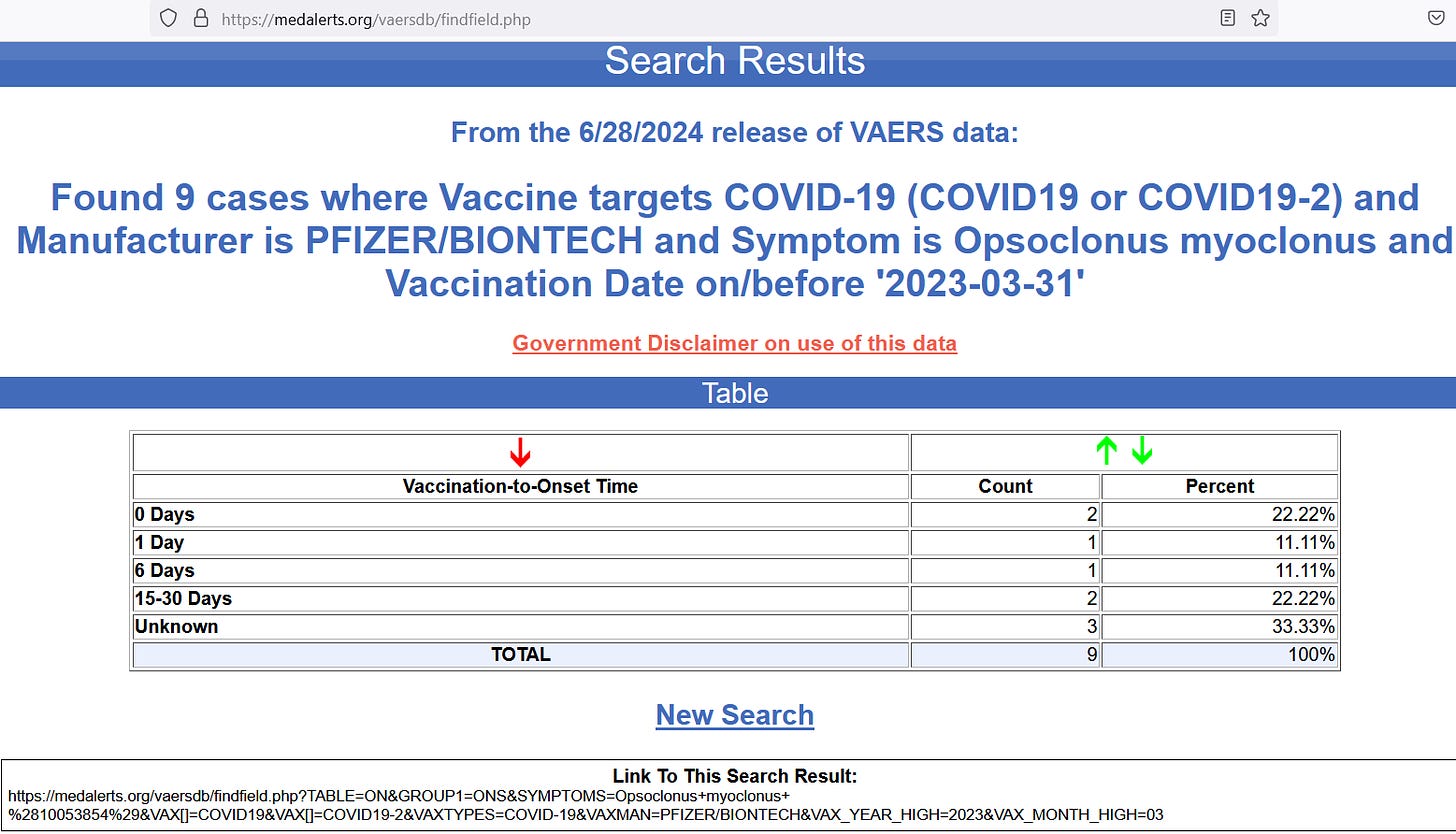
But when looking for adverse event reports in the VAERS system for Pfizer shot doses up to March 2023, there were 9 cases of it reported:
Six out of those 9 reports were known to occur within 30 days of the Pfizer shot injection, while data are unavailable for the time-window on the other 3 reports. But given how rare the background rate of incidence is for this disorder, having 9 cases show up should represent a red flag.
Past Red Flags
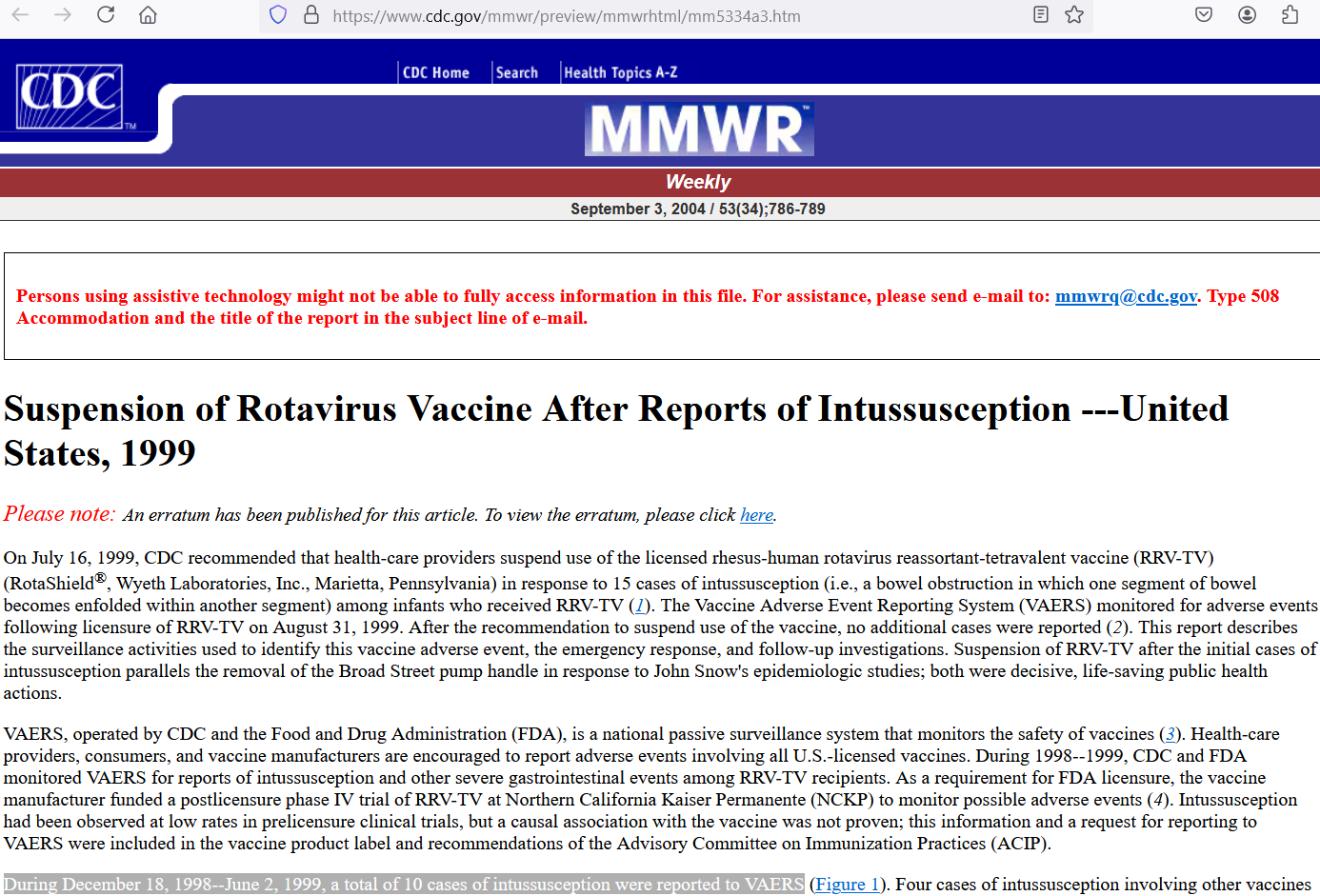
In the first half of 1999, regarding a newly-released rotavirus vaccine, RotaShield, there were 10 cases of intussusception (infolded intestines) in infants reported to the VAERS system — and just those 10 cases were already enough for the CDC to call for an investigation into the matter:
[click to enlarge]
But if 10 cases of intussusception were already enough to call for an investigation, then 9 cases of opsoclonus myoclonus should be enough to call for an investigation into Pfizer shots — because the background rate of intussusception is 1,700 times higher than the ultra-low background rate of opsoclonus myoclonus:
Extending the time-frame from the 6 months for RotaShield up to the “almost-30” months for Pfizer shots only scales you up by a factor of 5, but that is not enough to cover the 1,700-fold difference in background incidence rates — after reducing that difference by 5-fold, you still have a 340-fold difference to make up for by exposures.
Red-Flagged COVID shots
Another instance involves the COVID shots by Johnson & Johnson, leading to the CDC recommending a pause on the product after a total of 6 rare blood clots were reported. The rare condition is Thrombosis with Thrombocytopenia Syndrom (TTS) and it has a background yearly incidence rate of around 30 per 100,000.
Even those TTS cases involving unusual sites occur at a background incidence rate that is, at minimum, a full 76-fold higher background incidence rate than that of opsoclonus myoclonus:
[click to enlarge]
Evidence suggests that the Pfizer shot should be pulled from the market — and likely Moderna, too — because of exceeding the historic critical thresholds which have been utilized successfully in the past in order to pull dangerous products off the market. Considering how rare opsoclonus myoclonus is, the 9 reports to VAERS are alarming.
CDC recommended a pause after 6 cases of a disease that is at least 76 times more common than opsoclonus myoclonus, so 9 cases of opsoclonus myoclonus is serious.
Reference
[background rate of opsoclonus myoclonus is 0.02 yearly cases per 100,000] — Orphanet Report Series. https://www.orpha.net/pdfs/orphacom/cahiers/docs/GB/Prevalence_of_rare_diseases_by_decreasing_prevalence_or_cases.pdf
[background rate of intussusception is 34.2 yearly cases per 100,000] — Murphy TV, Gargiullo PM, Massoudi MS, Nelson DB, Jumaan AO, Okoro CA, Zanardi LR, Setia S, Fair E, LeBaron CW, Wharton M, Livengood JR; Rotavirus Intussusception Investigation Team. Intussusception among infants given an oral rotavirus vaccine. N Engl J Med. 2001 Feb 22;344(8):564-72. doi: 10.1056/NEJM200102223440804. Erratum in: N Engl J Med 2001 May 17;344(20):1564. Livingood, JR [corrected to Livengood, JR]. PMID: 11207352. https://www.nejm.org/doi/10.1056/NEJM200102223440804
[background rate of TTS is at least 1.53 yearly cases per 100,000] — Lloyd PC, Lufkin B, Moll K, Ogilvie RP, McMahill-Walraven CN, Beachler DC, Kelman JA, Shi X, Hobbi S, Amend KL, Djibo DA, Shangguan S, Shoaibi A, Sheng M, Secora A, Zhou CK, Kowarski L, Chillarige Y, Forshee RA, Anderson SA, Muthuri S, Seeger JD, Kline A, Reich C, MaCurdy T, Wong HL. Incidence rates of thrombosis with thrombocytopenia syndrome (TTS) among adults in United States commercial and Medicare claims databases, 2017-2020. Vaccine. 2024 Mar 19;42(8):2004-2010. doi: 10.1016/j.vaccine.2024.02.017. Epub 2024 Feb 22. PMID: 38388240. https://pubmed.ncbi.nlm.nih.gov/38388240/
[J&J vaccine pause suggested after just 6 cases in VAERS] — Scientific American. https://www.scientificamerican.com/article/blood-clots-and-the-johnson-johnson-vaccine-what-we-know-so-far/