Outsized share of certain Adverse Event Reports
The Proportional Reporting Ratios that CDC found
As Josh Guetzkow reported on, over at
, CDC finally let the cat out of the bag and released the safety signal data that they had agreed to be following each and every week. The report by Josh is both comprehensive and illuminating.The signalling process involves the share of all adverse event (AE) reports made up by a certain AE — such as the share of all reports made up by myocarditis. If all other vaccine products had 1% of all of their AE reports involve myocarditis — while 5% of the AE reports of a new product involve myocarditis — then that’d be a safety signal.
Having 5 times the share of your AE reports made up by a certain AE is “bad.”
The proportional reporting ratio (PRR) is this ratio or fraction:
[a specific event’s share of all AEs for product of interest]
——————————————————————————-
[that same specific event’s share of all AEs for all products]
Using the links Josh shared, I grabbed images and marked them up (as per usual). Here are the ones I selected, separated out by age groups.
Ages 5 to 11
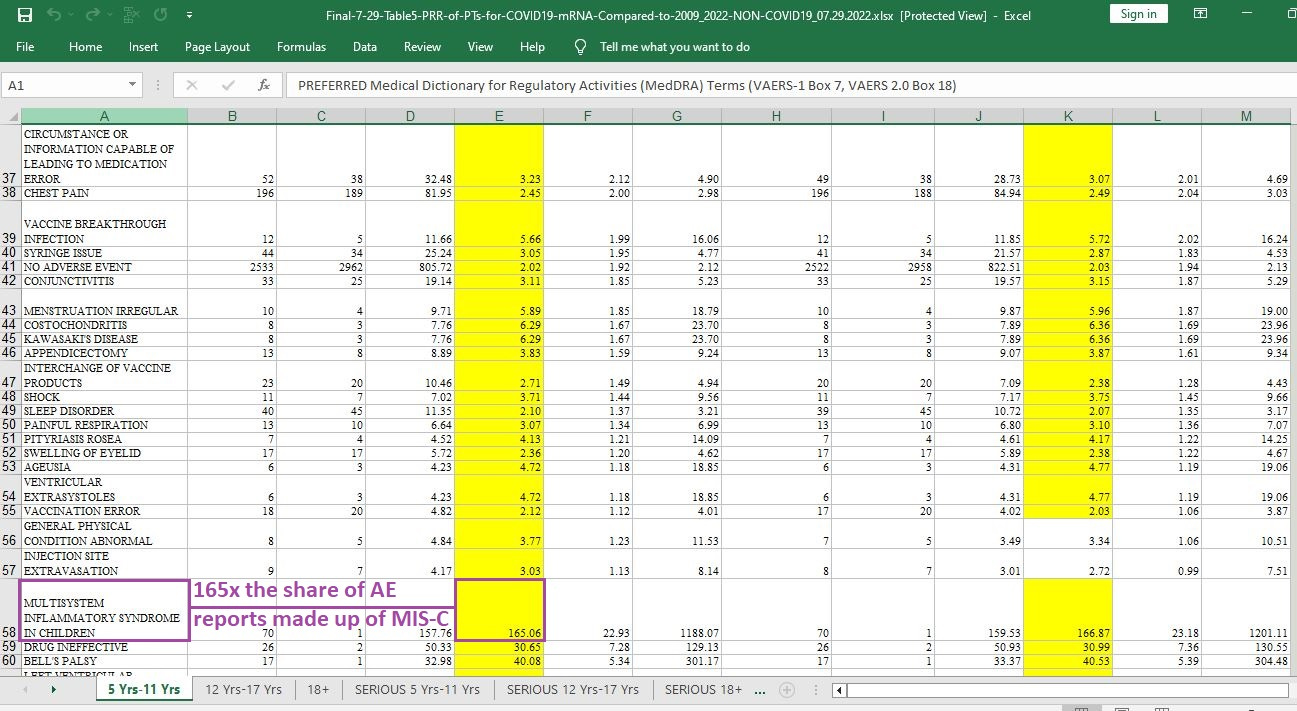
Multisystem (body-wide) inflammation in kids:
[click image to enlarge]
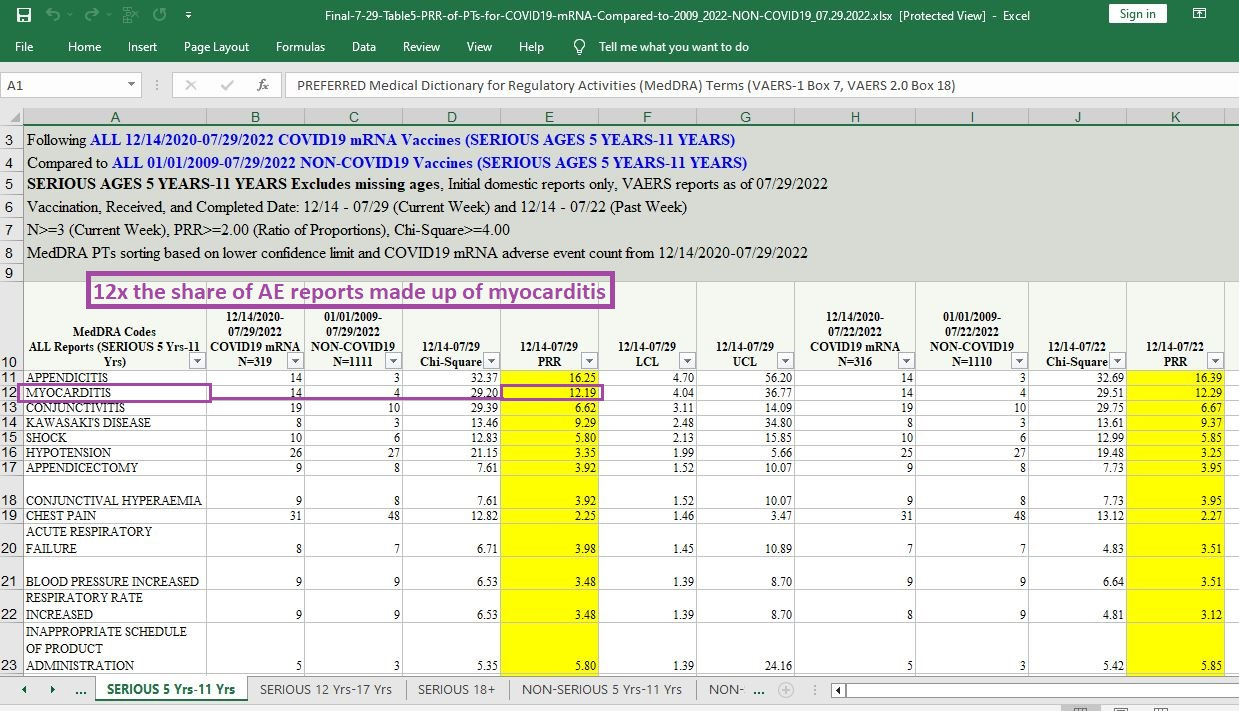
A dozen times the share of all AEs involving myocarditis:
[click image to enlarge]
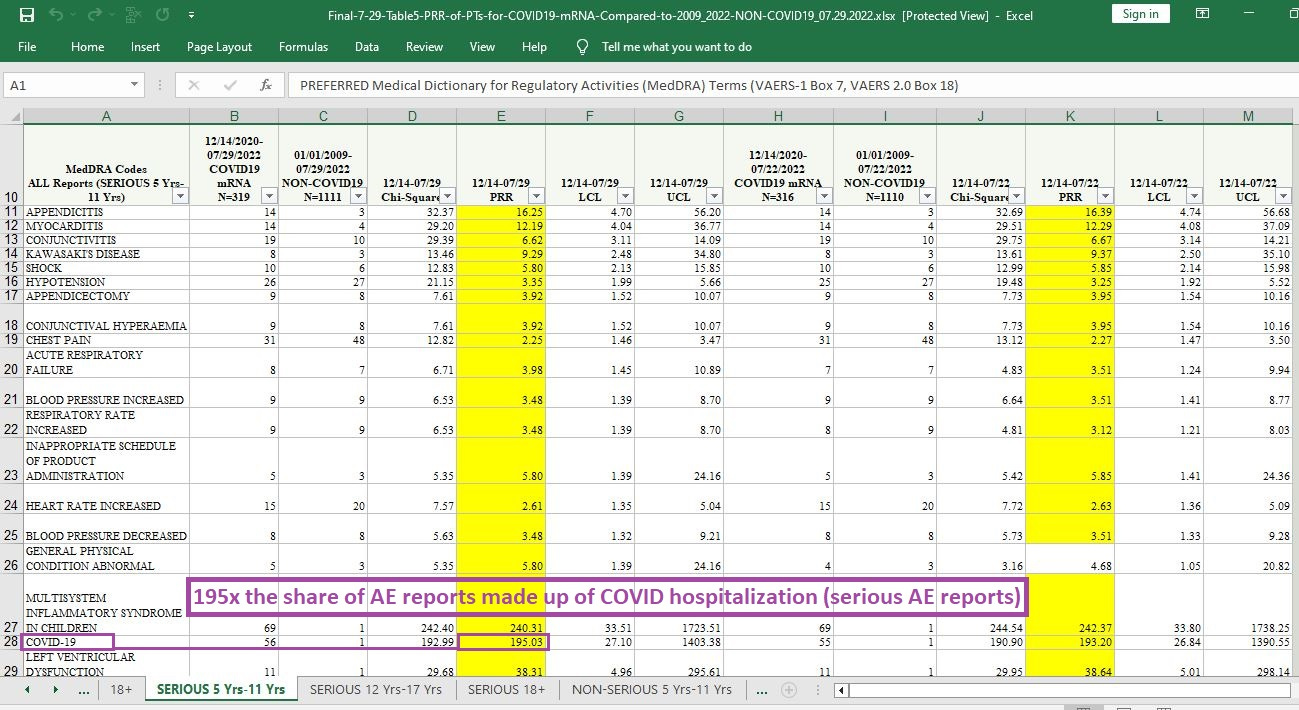
195 times the likelihood that a given AE report will involve COVID hospitalization or death — given that the COVID case is filed under “serious AE reports” (where, to be called serious, a minimum of hospitalization is required):
[click image to enlarge]
Ages 12 to 17
51 times the share of reports involving myocarditis:
[click image to enlarge]
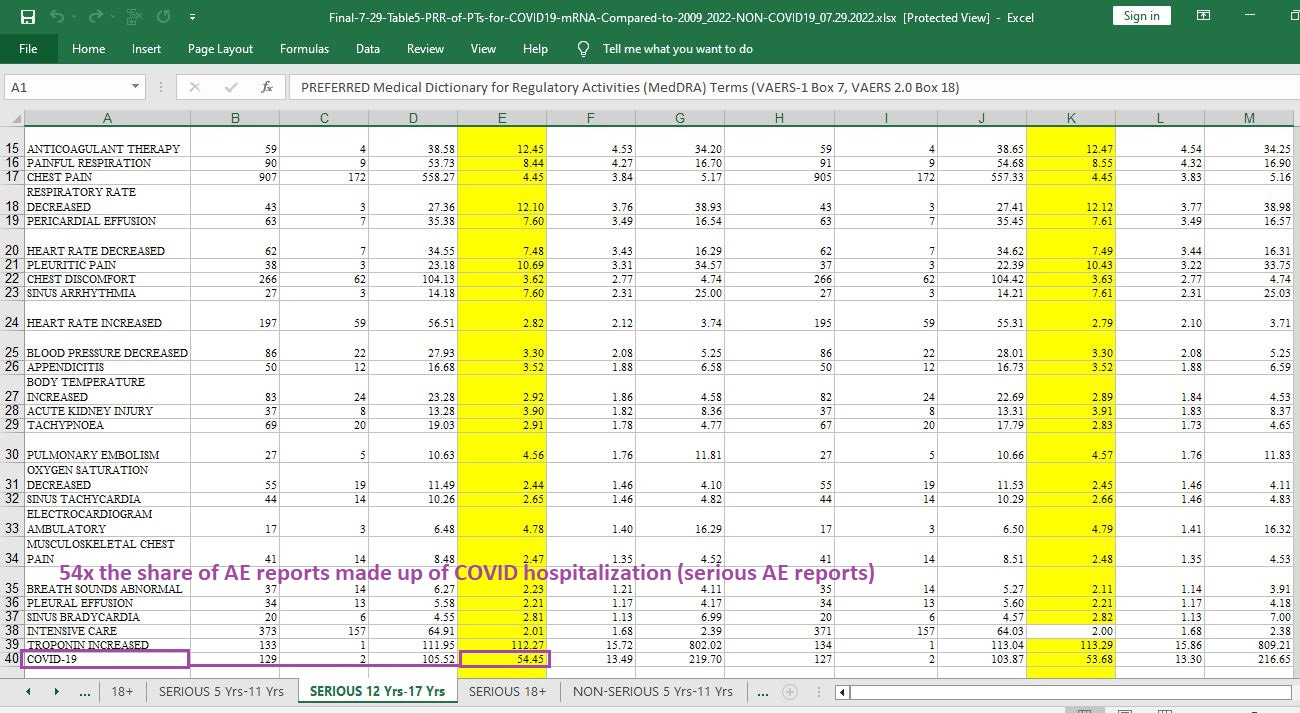
54 times the likelihood that a given AE report will involve COVID hospitalization or death — given that the COVID case is filed under “serious AE reports” (where, to be called serious, a minimum of hospitalization is required):
[click image to enlarge]
Age 18+
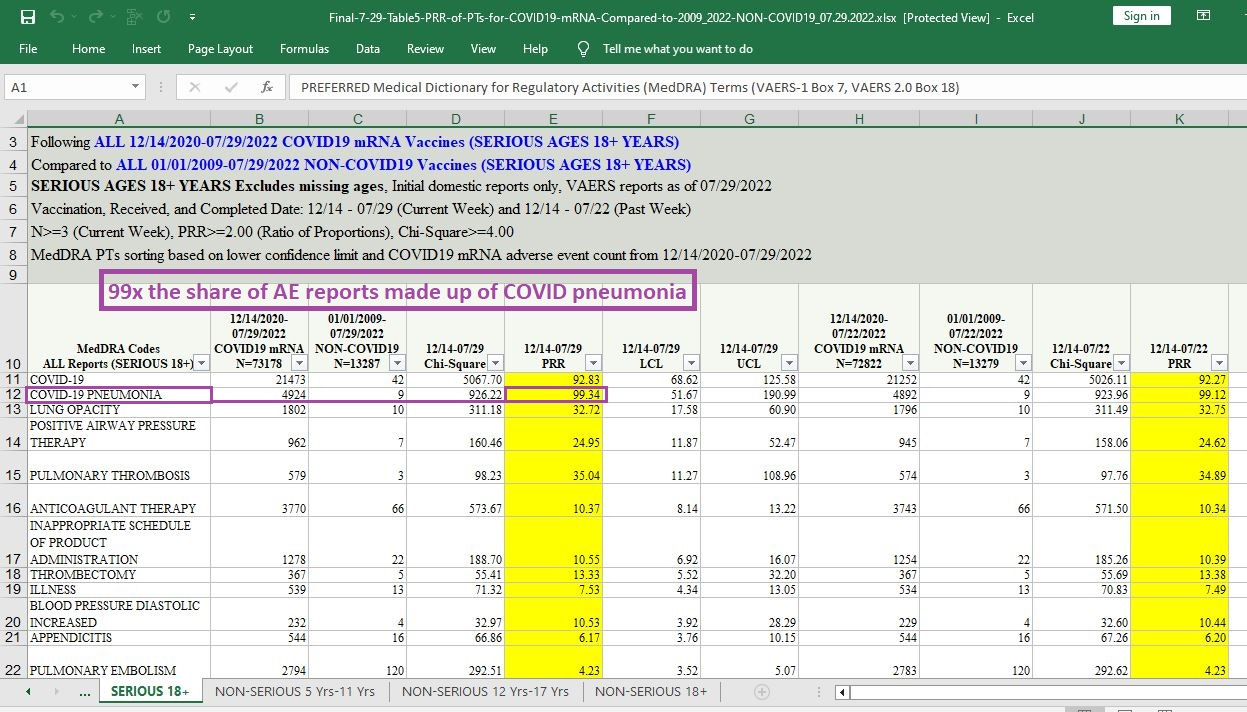
99 times the likelihood that a given AE report will involve hospitalization or death from COVID pneumonia — given that the COVID case is filed under “serious AE reports” (where, to be called serious, a minimum of hospitalization is required):
[click image to enlarge]
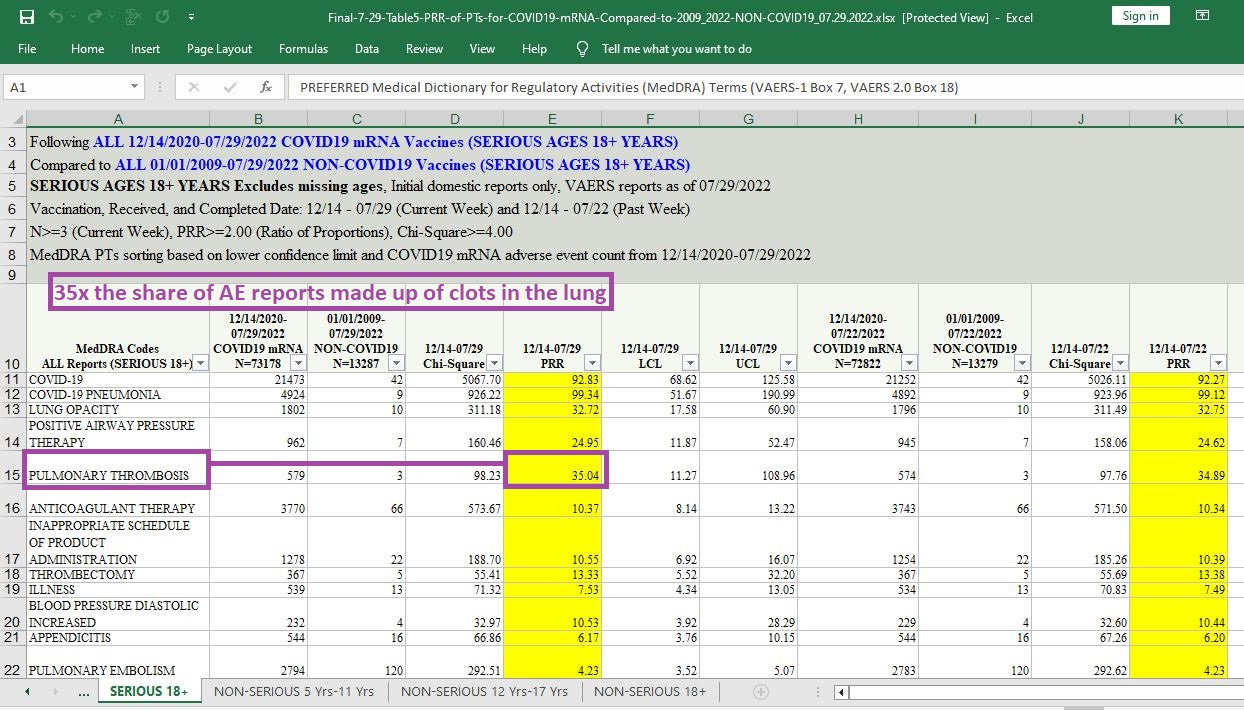
35 times the share of AE reports made up by clots in the lungs:
[click image to enlarge]
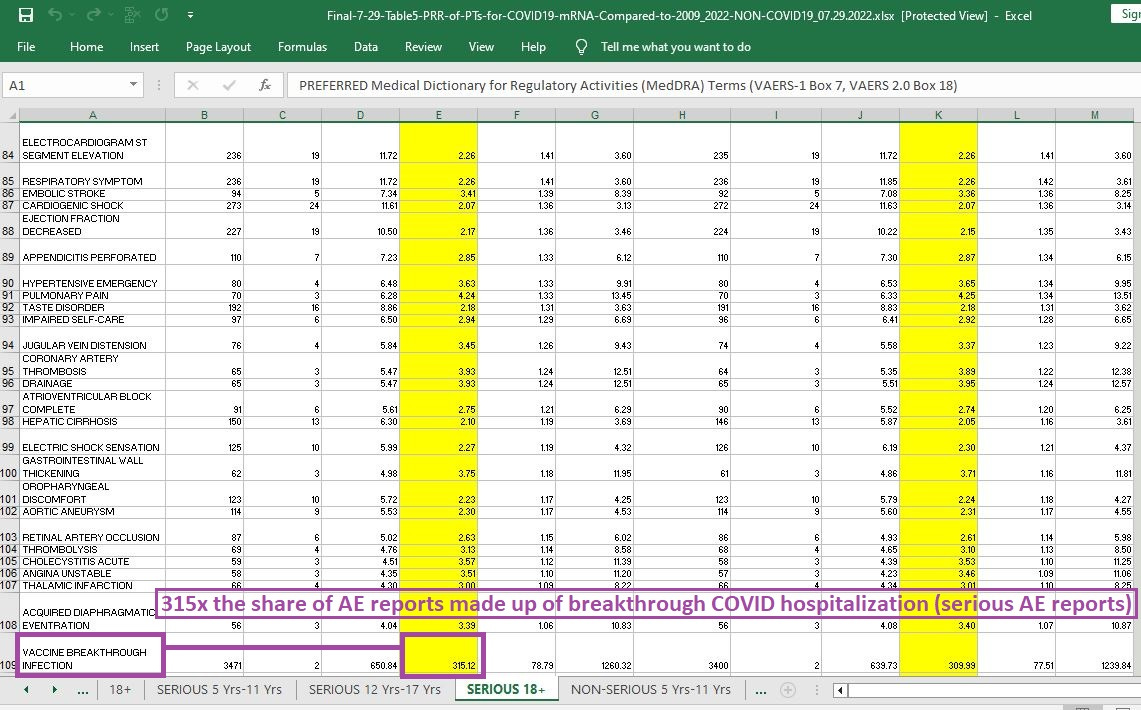
315 times the likelihood that an AE report will involve a breakthrough infection that you are hospitalized for:
[click image to enlarge]
If you looked at a random AE report for one of the mRNA shots and also for other vaccine products, it is 315 times more likely that the AE report for the mRNA shot involves a breakthrough infection so severe that it requires hospitalization.
The upshot:
Experimental COVID injections are a lot less safe, and a lot less effective, than typical vaccine products.