Relation between FDA and Drug Firms is too cozy to Work
The "what" and "who" using the Hepatitis B vaccine example
To know if something works requires knowing for “what” and for “who” it is working.
The winning odds that you get playing BlackJack at a casino work, but they do not work for you. Those odds work against you, but they work for the casino, allowing the casino to rake in loads of cash from people who play.
From lockdowns, masks, and COVID jabs, you can ask if they work. But the more important question is for what they are working, and for who.
One possibility is that things like COVID jab mandates work in order to line the pockets of vaccine billionaires, billionaires who have successfully captured the regulatory agencies that were supposed to police them.
An example of regulatory capture, where CEOs at drug firms get to decide which drugs make it to market — or at least get to restrict the review of their products so that bad things aren’t found — would be the approval of the Hepatitis B vaccine.
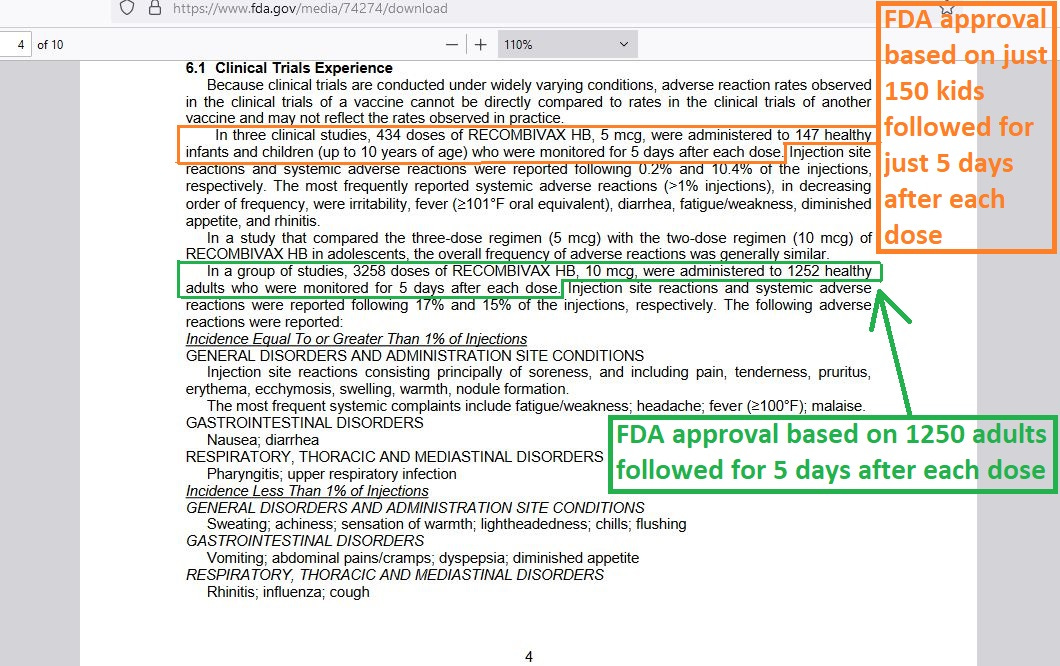
Here is an example, showing how it is that only 5 days of observation post-dose were considered by authorities to be sufficient to allow a drug firm access to a vaccine market:
But you can’t get adequate safety data from 5 days of observation. If you can’t get good data from it, then how come the FDA used it? The answer is simple: regulatory capture.
In a retracted study, Kostoff et. al. showed that you have to go out at least to Day 30 post-dose to capture the elevated death counts. That’s an absolute minimum, because experience with other vaccines shows some harms only showing up years after first introduction (vaccine for Dengue Fever causing severe Dengue 2 years later).
For what does the 5-day observation window work?
The restricted observation window works for expediting approval.
For who does it work?
It works in the interest of vaccine firms (and against the interests of public health) so that there is not enough observation time to discover bad effects of their products.
Reports reveal the fees from drug firms pay for 80% of all income of all federal regulators overseeing and approving those drugs. If FDA officials get 80% of their paychecks from drug firm fees, they cannot be trusted to remain objective.
This isn’t functionally different from criminals paying the salaries of the cops who police them.
Reference
[page 4 of package insert for Recombivax HB hepatitis B vaccine] — FDA. https://www.fda.gov/files/vaccines%2C%20blood%20%26%20biologics/published/package-insert-recombivax-hb.pdf
[vaccine for Dengue Fever causing severe Dengue 2 years after approval] — Editorial | Volume 18, ISSUE 2, P123, February 2018. The dengue vaccine dilemma. The Lancet Infectious Diseases. Published: February, 2018 DOI: https://doi.org/10.1016/S1473-3099(18)30023-9
[fees from drug firms cover 80% of the salaries of FDA officials] — Darrow JJ, Avorn J, Kesselheim AS. FDA Approval and Regulation of Pharmaceuticals, 1983-2018. JAMA. 2020 Jan 14;323(2):164-176. doi: 10.1001/jama.2019.20288. Erratum in: JAMA. 2020 Feb 11;323(6):573. PMID: 31935033. https://pubmed.ncbi.nlm.nih.gov/31935033/
Pull Quote:
… in 2018, user fees accounted for approximately 80% of the salaries of review personnel responsible for the approval of new drugs.”
[retracted Kostoff et al study showing elevated death reports for 30 days post dose] — Kostoff RN, Calina D, Kanduc D, Briggs MB, Vlachoyiannopoulos P, Svistunov AA, Tsatsakis A. Why are we vaccinating children against COVID-19? Toxicol Rep. 2021;8:1665-1684. doi: 10.1016/j.toxrep.2021.08.010. Epub 2021 Sep 14. Retraction in: Toxicol Rep. 2022 May 06;9:1065. Erratum in: Toxicol Rep. 2021;8:1981. PMID: 34540594; PMCID: PMC8437699. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8437699/