State-of-the-Science when CDC advised stopping a vaccine in 1999
What they knew, and when they knew it
In mid-July of 1999, the CDC recommended a halt to the administration of a rotavirus vaccine called RotaShield. It is of interest to find out what they knew when they advised the halt in administration of RotaShield vaccine to infants.
Their report mentions their data, and you can go back and develop best-case and worst-case scenarios — identifying what level of known risk it took to get a vaccine pulled from market.
True risk would be discovered later by future researchers looking back in time on the cases of a serious adverse event in infants, intussusception (an infolding of the intestine), linked to RotaShield vaccine.
But this Substack bypasses the nuance of how high actual risk actually was, and instead it shows what “actionable risk” looks like.
When contrasted against known risk of the COVID shots, Moderna in particular, it can be proven that known risk is twice as high as the actionable risk which got RotaShield pulled.
Sources:
MMWR July 16, 1999 report (CDC) & PMC9021987 (4-nation myocarditis study)
As you can see, Nordic researchers found myocarditis in young males at twice the rate that it took to get a vaccine pulled from market back in 1999, though critics and detractors may try to argue that intussusception is more dangerous than myocarditis.
Even if they were able to prove that intussusception was worse (which is going out on a limb), there are still other risks such as stroke which are found from COVID shots that would prove that COVID shots are more dangerous than RotaShield was.
8 is Enough
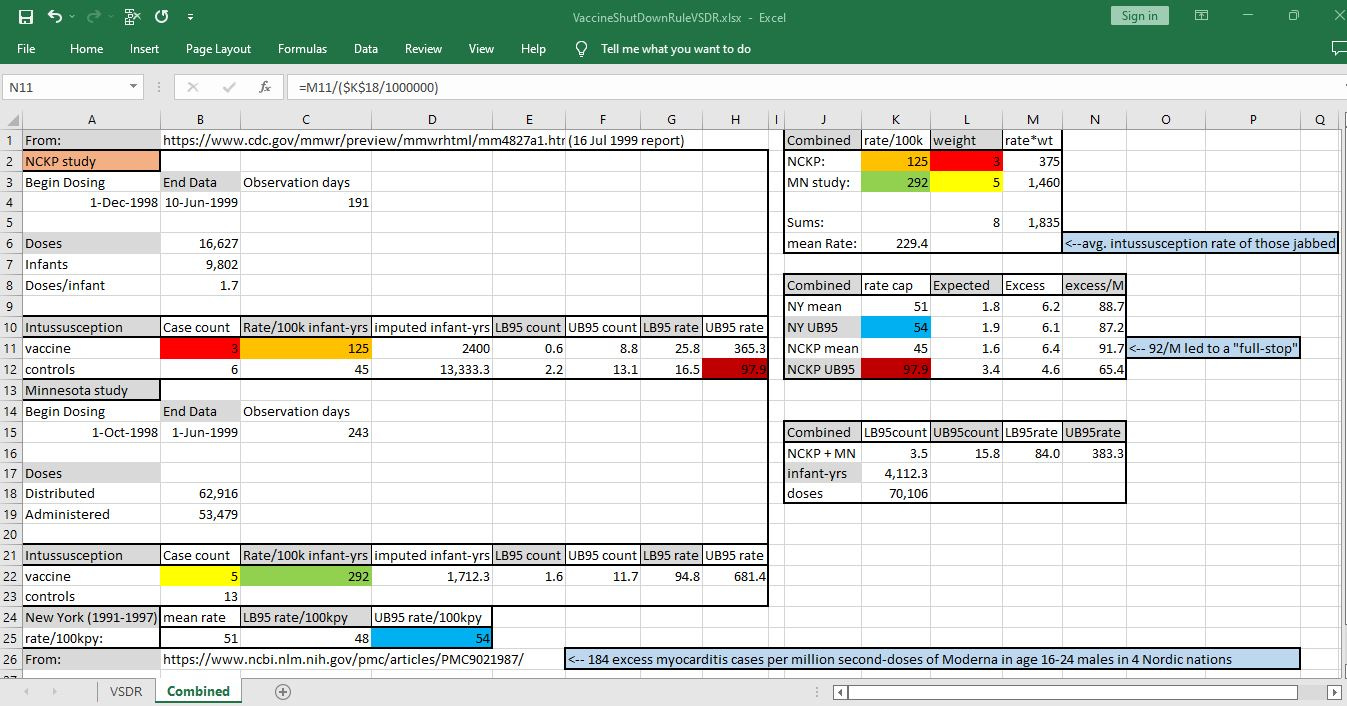
By mid-July of 1999, when they called for a halt on the RotaShield vaccine, CDC knew about 15 spontaneous reports of intussusception in VAERS and also they knew about another 8 case from post-market surveillance studies.
The 8 cases found could be combined together and compared against one of 4 possible estimates of background rates of intussusception — all stemming from that 16 Jul 1999 report. Here are the notes showing how:
[click on image above to enlarge it]
CDC uses two possible cutoff values for determining whether there is an excess in a rate: the historic average rate and the upper bound on a 95% prediction interval.
In the report calling for a halt to RotaShield, there were two possible averages — one from Northern California Kaiser Permanente (NCKP) surveillance, and one from 7 years of data for the state of New York.
These two sets of data can also be used in order to find the upper bound of a 95% interval. While prediction intervals that CDC uses are just a little bit larger than traditional confidence intervals are, the upper bound on a 95% confidence interval serves as a good estimate.
Deep Stats
The upper bound for background intussusception rates in New York was given in the July 1999 CDC report. The upper bound for the control group in the NCKP study was computed using CDC’s preferred method, “gamma method”, where you get lower and upper bound counts in a Poisson process by using an inverse standard gamma function.
Each background estimate leads to a different estimate in the excess intussusception related to vaccine use. The lowest estimate of background rate (the rate of intussusception in controls in NCKP study) was used in order to show the worst-case scenario that CDC officials had before them, when they called for a halt.
Future research would reveal that that worst case scenario (92 excess intussusception cases per million vaccine doses) was the most agreed-upon by researchers, and it represents one excess intussusception case for each 11,000 doses administered.
If a vaccine causes a serious adverse event for every 11,000 doses (91 serious adverse events per million doses), the precedent suggests that that vaccine should be pulled — because more than 90 serious AEs per million doses is “too much” (unacceptable) harm from a vaccine.
Other and safer alternatives should be used in place of vaccines that cause serious harm at such a high rate.
Reference
[CDC report calling for a stop to the administration of RotaShield vaccine after analysis of 8 cases] — CDC. Intussusception Among Recipients of Rotavirus Vaccine -- United States, 1998-1999. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4827a1.htm
[Follow-up research revealing a best-estimate intussusception risk of 1 new case per 11,000 doses of RotaShield vaccine] — Glass RI, Bresee JS, Turcios R, Fischer TK, Parashar UD, Steele AD. Rotavirus vaccines: targeting the developing world. J Infect Dis. 2005 Sep 1;192 Suppl 1:S160-6. doi: 10.1086/431504. PMID: 16088799. https://pubmed.ncbi.nlm.nih.gov/16088799/
[Nordic study showing myocarditis rates in young males of up to 184 per million doses] — Karlstad Ø, Hovi P, Husby A, Härkänen T, Selmer RM, Pihlström N, Hansen JV, Nohynek H, Gunnes N, Sundström A, Wohlfahrt J, Nieminen TA, Grünewald M, Gulseth HL, Hviid A, Ljung R. SARS-CoV-2 Vaccination and Myocarditis in a Nordic Cohort Study of 23 Million Residents. JAMA Cardiol. 2022 Jun 1;7(6):600-612. doi: 10.1001/jamacardio.2022.0583. PMID: 35442390; PMCID: PMC9021987. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9021987/