Vaccine effectiveness (VE) is the percentage of risk reduction you get compared to the untreated, usually aimed at the comparative incidence of symptomatic infection.
The FDA standards for acceptable and unacceptable levels of vaccine effectiveness have lowered for the COVID era. It used to be the case that the lower bound on a 95% confidence interval around the central estimate of vaccine effectiveness was required to be above 50% VE.
Here is an example of prior FDA standards:
[click on image to enlarge]
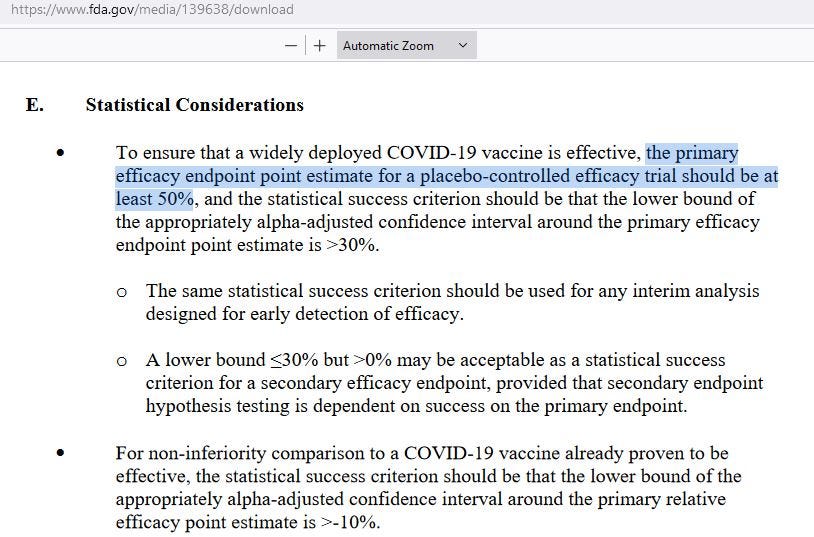
The new and lower standard for COVID injections was a central estimate of 50% VE, along with a 95% lower bound that was greater than 30% VE. At 30% VE, you get infected at a rate that is 70% as high as someone who remains unvaccinated.
Here is the “new” FDA standard for COVID injections:
[click on image to enlarge]
And here is how the two standards look when compared together on the same scale:
[click image to enlarge it]
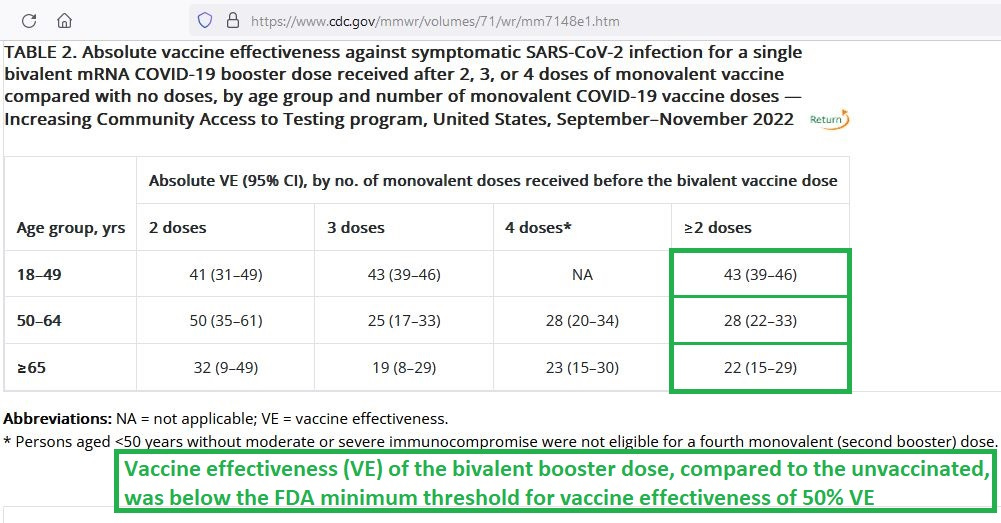
But when the new-fangled, bivalent booster was tested, it did not measure up to FDA standards:
[click image to enlarge it]
Neither the original monovalent “vaccine” — nor the bivalent booster — have the required effectiveness against Omicron to qualify for Emergency Use Authorization, let alone for FDA Approval.