When 23 Serious Adverse Events got a Vaccine Pulled from Market
RotaShield in 1999
NOTE: This post has now been edited to reflect the true per-each-dose rate of serious AEs found in Fraiman et. al.
RotaShield was a rotavirus vaccine that led to a serious adverse event (AE) called intussusception, where the intestine folds inside of itself as when you fold a pair of socks together, one over the other. Surgical resection is often required.
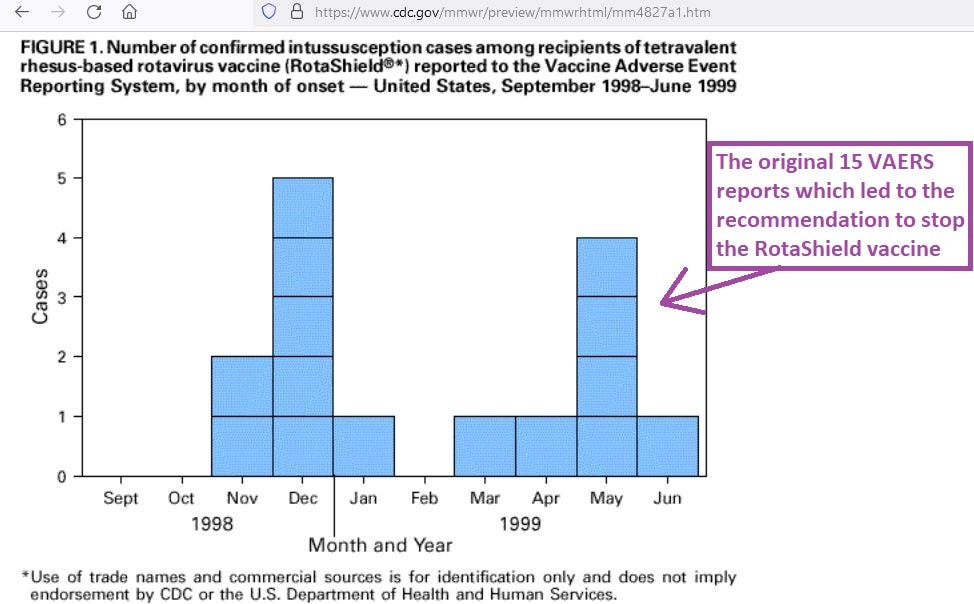
After 15 total VAERS reports had been made about intussusception in infants vaccinated with RotaShield over the course of about 6 months, the CDC recommended a halt to the administration of it. Here are the months when those 15 reports were made:
There were actually 23 known cases of intussusception after RotaShield when CDC determined that the safety signal was strong enough to issue a recommendation in Morbidity & Mortality Weekly Report (MMWR) to halt its administration to infants.
Along with the estimated 1.5 million doses which had been given, those 23 cases were occurring at a rate of 15.3 intussusception cases per million vaccine doses administered.
And reports kept coming in after the recommendation to halt, even though all of the remaining reports for the year were in infants vaccinated by 17 Jul 1999 — around the time when the vaccine manufacturer voluntarily removed the RotaShield vaccine product from market:
Two post-market surveillance studies were reported by CDC at the time, one by Northern California Kaiser Permanente (NCKP) and another study in Minnesota.
The NCKP study was inconclusive because of not having enough occurrences of the event of interest (they had 3 vaccine-associated intussusception cases). The Minnesota study appears a little more conclusive of vaccine-associated harm (they had 5 cases).
CDC’s reporting of the details are sparse but it was possible to piece together the “state-of-the-science” at the time, using an assumption of non-heterogeneity and an advanced method for finding statistical significance from the overlap of confidence intervals on the rates of intussusception experienced in those who took the vaccine.
Deep Stats
[click on the image above to enlarge it]
In just over 6 months, the control group for the NCKP study had 13,000 total infant-years of observation, which is large enough to develop at least a rough indication of what the “natural” or background rate of intussusception is in the unvaccinated infant population.
Any intussusception rates between 20 and 90 cases per 100,000 infant-years would be deemed normal under the limited data, but rates outside of that range would not.
That range results from the choice to run a 90% confidence interval around the rates of both groups (vaccine and control) in order to perform a Confidence Interval Overlap test which was set specifically to keep the significance level at 0.05 or below (a 5% chance of overlap).
The setting of the confidence level requires knowing the ratio of standard errors between groups, so as to make sure that the probability of any overlap is 5% (0.05) or below. Using the NCKP control group as the benchmark confidence interval, the Minnesota intussusception rates were significantly higher than expected.
The lower bound of the interval for Minnesota intussusception rates in those vaccinated was even higher than the upper bound of the interval for controls — albeit controls from another study.
The upshot is that CDC recommended a complete shut-down of a vaccine based on reports of just 23 cases of a single kind of serious AE, across 1.5 million doses (15.3 serious AEs per million doses).
Contrast that against a re-analysis of the Phase III trials for COVID shots, which found an excess of 625 serious AE's per million doses.
That’s 41 times the rate of serious AE’s that it took to shut-down a previous vaccine in 1999. If 15.3 serious AE’s per million doses was enough to get a vaccine pulled back then, how come 41 times as many serious AE’s isn’t enough regarding the COVID shots?
Are medical regulators 41 times less risk-averse than they were two decades ago?
Speeding Ticket Analogy
Using the analogy of speeding in your car, if going 10 mph over the speed limit was enough to get you a speeding ticket back in 1999 from the regulators, or at least a warning from them, then how come going 410 mph over the speed limit today isn’t enough?
By how many more multiples do things have to get worse, before regulators step in?
Reference
[July 16, 1999 report on potential danger of RotaShield vaccine] — CDC. https://www.cdc.gov/mmwr/preview/mmwrhtml/mm4827a1.htm
[CDC review of VAERS data which mentions RotaShield] — CDC. https://www.cdc.gov/mmwr/preview/mmwrhtml/ss5201a1.htm
[Reanalysis of the Phase III Clinical Trials for mRNA COVID shots, revealing 625 serious adverse events per million doses] — Fraiman J, Erviti J, Jones M, Greenland S, Whelan P, Kaplan RM, Doshi P. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine. 2022 Sep 22;40(40):5798-5805. doi: 10.1016/j.vaccine.2022.08.036. Epub 2022 Aug 31. PMID: 36055877; PMCID: PMC9428332. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9428332/
[How to set confidence levels based on the ratio of two standard errors, so that you end up with a 5% chance of confidence interval overlap (Table 3)] — Payton ME, Greenstone MH, Schenker N. Overlapping confidence intervals or standard error intervals: what do they mean in terms of statistical significance? J Insect Sci. 2003;3:34. doi: 10.1093/jis/3.1.34. Epub 2003 Oct 30. PMID: 15841249; PMCID: PMC524673. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC524673/