Key:
NNV: number needed to be vaccinated to prevent something
NNH: number needed to be vaccinated to cause a harm
NNV-Hosp: number needed to be vaccinated to prevent a single hospitalization
NNH-Hosp: number needed to be vaccinated to cause an extra hospitalization
In this prior Substack, I looked at an absolute risk of COVID hospitalization versus one estimate of risk of hospitalization due to serious adverse events from the COVID jab. But this Substack uses more robust estimates from both the CDC and from a reanalysis of Phase III clinical trial data.
NNV-Hosp
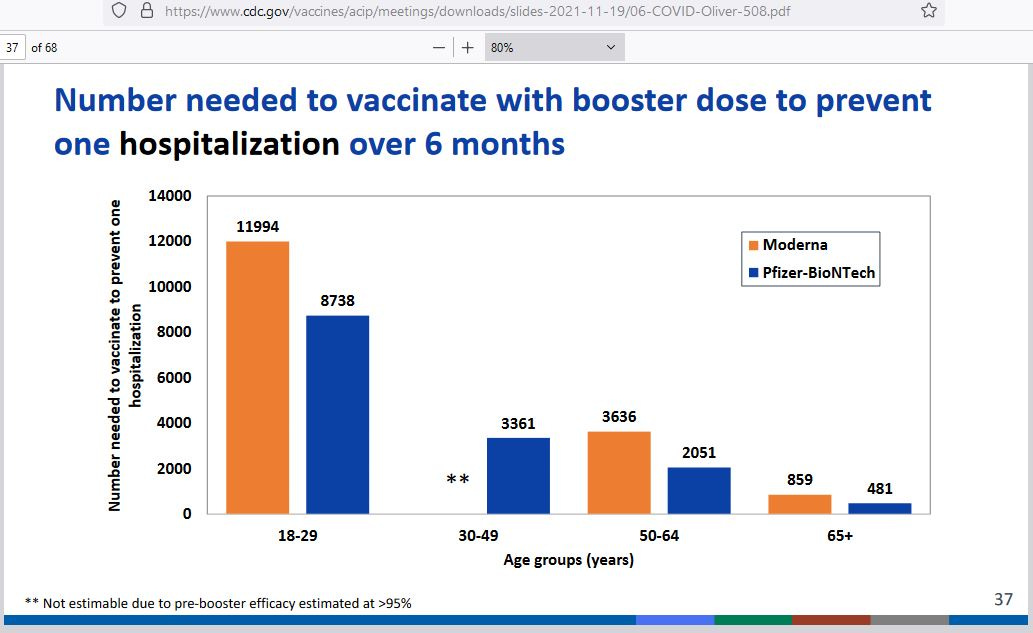
Here is the CDC estimate regarding the number of booster shots that must be given out in order to prevent a single COVID hospitalization among age groups:
To get a broad estimate for each age group, I combined/averaged the Moderna and Pfizer numbers.
NNH-Hosp
In a reanalysis of Phase III clinicial trials, it was discovered that — when data from both Moderna and Pfizer got combined, there was an excess of serious “adverse events of special interest” (AESI) — the type that cause hospitalizations — of 12.5 per 10,000 recipients of the jab.
That combined estimate covered an absolute time window of 4 months, and the median length of follow-up for participants in the trial was 2 months or more. That means that there were 12.5 excess serious AESI’s per 20,000 person-months.
If it is assumed that that risk remains stable for 6 months of follow-up, then a converted estimate for 10,000 persons followed for 6 months would be:
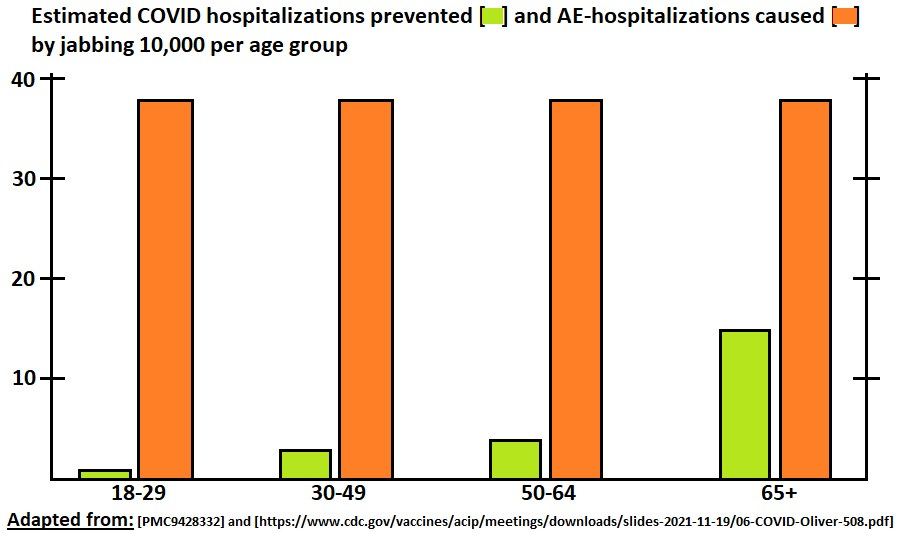
38 serious AESI per 10,000
Here is how it looks when presented side-by-side per age group, with prevented hospitalizations in lime green at left each time, and the expected extra hospitalizations caused by adverse events in orange on the right:
Using this estimate, in no age group is it expected for COVID jabs to prevent more hospitalizations than they cause.
This estimate may be a rough estimate, but there is also an excellent review (cited below) showing how, after August 2021 (i.e. Delta variant), the NNV-Hosp was at least 2,000 regardless of location.
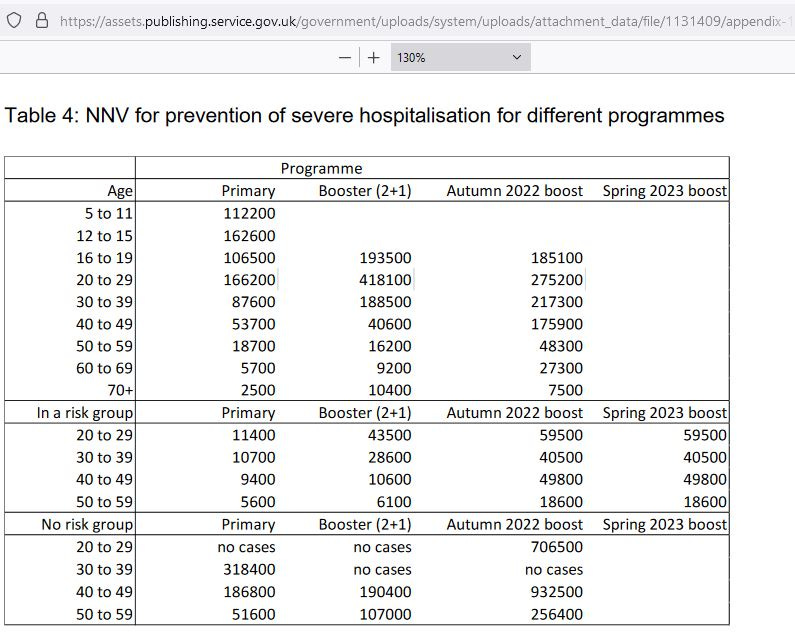
Even worse, there are data from the UK showing an NNV-Hosp of over 10,000 for those below middle-age. If one in 800 are hospitalized for serious adverse events, that means 12.5 go into the hospital for every one that is kept out.
The risk:benefit profile for COVID booster shots is horrendous. Who, in their right mind, would want to take a shot that causes over 12 adverse event hospitalizations for each COVID hospitalization that it prevents?
How far must a government regulator have his or her head in the sand to not notice this? It calls into question whether government regulatory institutions are now completely compromised by those with adversarial interests.
What else could explain a regulatory “oversight” that is this large in magnitude?
Reference
[excellent review showing how it now requires at least 2,000 “vaccinations” to prevent a single COVID hospitalization; regardless of location] — Larkin A, Waitzkin H, Fassler E, et al. How missing evidence-based medicine indicators can inform COVID-19 vaccine distribution policies: a scoping review and calculation of indicators from data in randomised controlled trials. BMJ Open 2022;12:e063525. doi: 10.1136/bmjopen-2022-063525. https://bmjopen.bmj.com/content/12/12/e063525
[more than 10,000 booster shots required to prevent a single hospitalization in those below middle age] — Appendix 1: estimation of number needed to vaccinate to prevent a COVID-19 hospitalisation for primary vaccination, booster vaccination (3rd dose), autumn 2022 and spring 2023 booster for those newly in a risk group. Based on a UK Health Security Agency (UKHSA) presentation to the Joint Committee on Vaccination and Immunisation (JCVI) on 25 October 2022. https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/1131409/appendix-1-of-jcvi-statement-on-2023-covid-19-vaccination-programme-8-november-2022.pdf
The lowest NNV for boosters against “severe” hospitalization above was 7,500:
[at least 1 serious AESI per 800 recipients when trials are combined] — Fraiman J, Erviti J, Jones M, Greenland S, Whelan P, Kaplan RM, Doshi P. Serious adverse events of special interest following mRNA COVID-19 vaccination in randomized trials in adults. Vaccine. 2022 Sep 22;40(40):5798-5805. doi: 10.1016/j.vaccine.2022.08.036. Epub 2022 Aug 31. PMID: 36055877; PMCID: PMC9428332. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9428332/
[CDC slide showing 10,000 booster shots are required to prevent a single COVID hospitalization in those under age 30] — CDC Presentation. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-11-19/06-COVID-Oliver-508.pdf